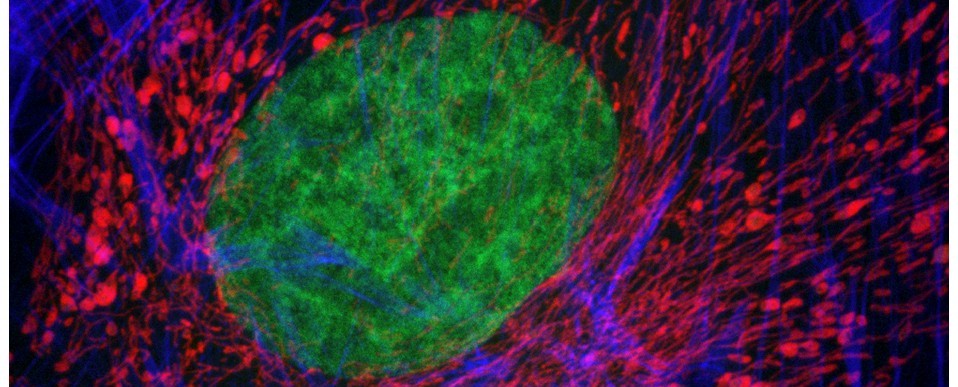
Above: Photo-bleaching (fading) occurs when a fluorochrome permanently loses the ability to fluoresce due to photon-induced chemical damage and covalent modification.
Hands up if you’ve spent hours preparing a sample for fluorescence microscopy only to see the signal disappear before your eyes upon excitation? Frustrating eh (unless, of course, FRAP is your objective)? Well here’s some simple and sound advice on how you can minimise photo-bleaching and get the best out of your samples under the fluorescence microscope.
1. Visualise your samples immediately after fluorescent labelling – this is when they are at their brightest.
- If this is not possible then loosely wrap your samples in aluminium foil and keep them in the dark at 4oC until you get the opportunity to image them.
2. Minimise their exposure to light in order to reduce photo-bleaching.
- visualise your samples under low light conditions.
- use transmitted light to find a region of interest (ROI) and then switch to epifluorescence observation – avoid dwelling too long on the ROI.
- step down the intensity level of excitation light or insert a neutral density filter into the light path.
- set up imaging parameters on a neighbouring region and then return to the ROI for image capture.
- use image binning to reduce exposure time.
- use the microscope shutter to switch off the light source between images.
- create a photo-bleach curve from a timed series of images. This can be used to normalise for loss of fluorescence intensity.
3. Switch to a mounting medium with anti-fade protection e.g. Vectashield, Prolong Gold/Diamond, SlowFade Gold/Diamond. These work by reducing the oxygen available for photo-oxidation reactions, thus reducing photo-bleaching. N.B. Many of these are available with a nuclear counterstain (e.g. Dapi) included in the formulation. Alternatively, make your own anti-fade reagent (instructions below).
4. Switch to brighter, more photo-stable fluorochromes. First generation fluorochromes such as FITC and TRITC photo-bleach readily (and are pH sensitive) thus should be replaced with modern dyes such as the Alexa Fluor, Dylight, or Atto range of fluorochromes, which are much brighter and far more photo-stable.
Good luck!
AJH
Further reading
- Wright Cell Imaging Facility: Mounting Media and Anti-fade Reagents
- Photobleaching: a review
![[Parameter-Settings] FileVersion = 2000 Date/Time = 0000:00:00 00:00:00 Date/Time + ms = 0000:00:00,00:00:00:000 User Name = TCS User Width = 1032 Length = 1032 Bits per Sample = 8 Used Bits per Sample = 8 Samples per Pixel = 3 ScanMode = xy Series Name = demo2.lei](http://blogs.cardiff.ac.uk/bioimaging/wp-content/uploads/sites/492/2016/09/Photobleach-1024x509.jpg)