Rheumatoid Arthritis Awareness Week: Our Research
3 September 2020
Rheumatoid Arthritis Awareness Week
Rheumatoid Arthritis Awareness Week takes place on 7-11 September 2020 and is organised by the National Rheumatoid Arthritis Society (NRAS). The Centre for Trials Research is active in a number of studies to help inform evidence base in this important area. There is a focus on wellbeing and there are multiple events running through the week, which can be found here https://www.nras.org.uk/raaw2020
What is Rheumatoid Arthritis?
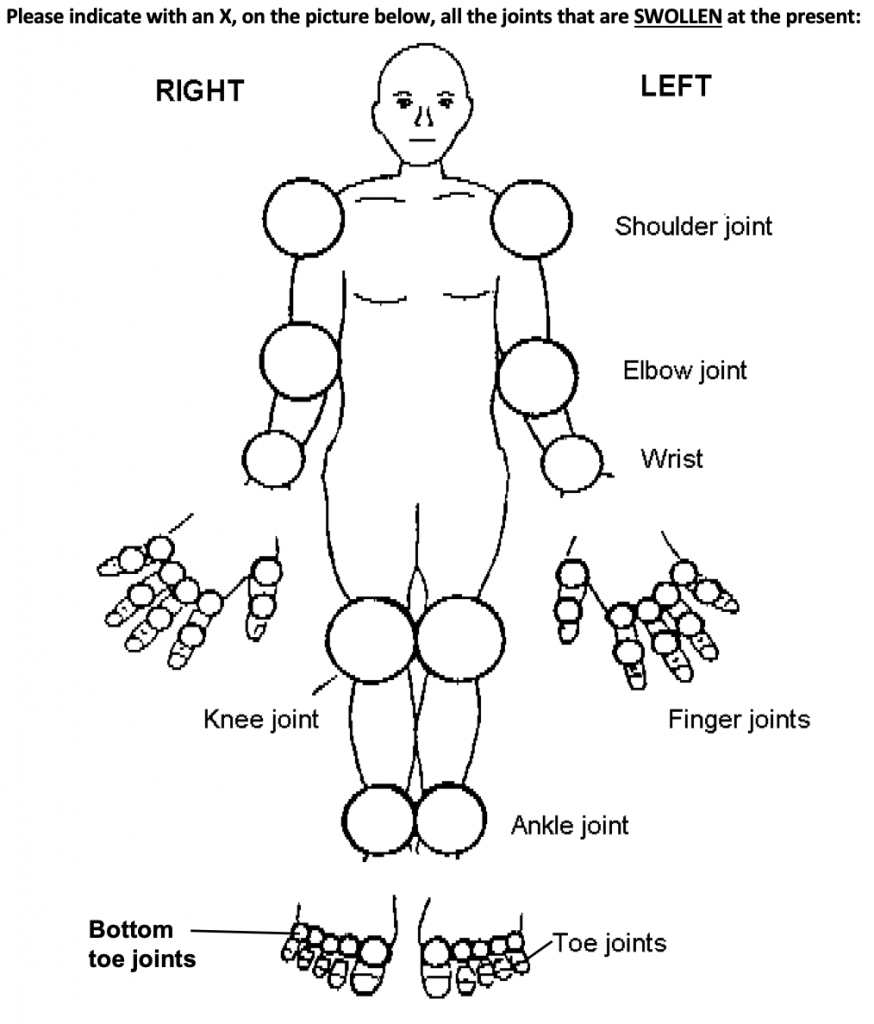
Rheumatoid Arthritis (RA) is a chronic autoimmune disease in which the body’s immune system attacks the joints, bones and cartilage. As with many autoimmune diseases, the cause of RA has not yet been determined; it is assumed that there may be certain genetic and environmental factors associated.
Effects of RA
Symptoms of RA include warm, swollen, and painful joints commonly in the wrist and hands but possible throughout all joint areas and on both sides of the body.
RA is characterised by fluctuating symptom severity, differential disease activity and recurrent flares.
There is no known cure for Rheumatoid Arthritis, so management and treatment modifications are key. Regular monitoring is absolutely vital but patients need to be assessed by doctors or nurses using composite measures involving blood tests, clinical assessment and patient-reported outcomes.
Who Suffers from RA?
RA affects about 1% of the UK population. Women are three times more likely than men to have this disease and onset is possible from any age, often 40-60.
Getting Involved in CREATE
I began working in the Cardiff Regional Experimental Arthritis Treatment and Evaluation Centre (CREATE) Centre with Professor Ernest Choy in 2014. The CREATE Centre is joint funded by Versus Arthritis (formerly Arthritis Research UK) and Health and Care Research Wales.
Involvement with CREATE
My role focussed on early phase clinical trials of treatments for Rheumatoid Arthritis and determining how adaptive trial designs could be implemented. I undertook a systematic review of all early phase clinical trials in RA to uncover if any adaptive designs had already been implemented and also determine the average number of patients and arms in these trials to provide an estimate of how efficient an adaptive design would need to be in these situations to make their implementation worthwhile. I also looked into the trial design, use of blinding and randomisation, and also the most used primary outcomes and at what point in the trial these where measured. This was published in Rheumatology Advances in Practice.
Professor Choy and I also created a Special Interest Group on Adaptive Design Trials within the OMERACT (Outcome Measures in Rheumatology) Initiative, which included members from academic, clinical and private industry in the UK, Germany, The Netherlands, the USA, Denmark and Hong Kong. This group aided with the writing with the systematic review and we met regularly over the phone for a couple of years in the lead up to the OMERACT 2018 Conference. I wrote a report of the Special Interest Group progress for the proceedings of the conference, which was published in the Journal of Rheumatology.
Involvement with SOCRATES

For some time, I’d held an idea that I could do a Fellowship, specifically one through which I could obtain a PhD, based around Rasch measurement theory (RMT) and the development and validation of Patient Reported Outcome Measures (PROMs). This because I had undertaken a part-time MSc dissertation within CTR on Rasch measurement theory using PROMs data from the DEPICTED trial, and enjoyed implementing RMT.
The systematic review above, reading around the subject and discussions with Professor Choy, led me to discover that there was scope to apply RMT to measures of symptom severity in Rheumatoid Arthritis. Through training courses and discussions with further potential supervisors, I realised the possibility of developing a computer adaptive test, which could help with many other issues that exist within RA, such as the lack of regular monitoring.
NIHR Doctoral Fellowship
I therefore set about applying for NIHR Doctoral Fellowship with Professor Ernest Choy as my primary supervisor and with Dr Mike Horton (University of Leeds), Dr Karl Bang Christensen (University of Copenhagen), Dr Rhiannon Phillips (Cardiff Metropolitan University) and Dr David Gillespie (Cardiff University) in the supervisory team. I submitted ‘Patient-reported Outcome Measures of Rheumatoid Arthritis Symptom Severity: development of a computer adaptive test from an item bank using Rasch measurement theory (SOCRATES)’ in December 2018 and, following success at interview, began in October 2019. The fellowship is funded by Health and Care Research Wales (NIHR-FS-19).
The NIHR Doctoral Fellowship is both a research and training fellowship and I was lucky to have many training courses lined up straight away whilst I set about the process of setting up data collection, which involved collecting data from the items of all existing relevant PROMs in this field discovered from a systematic search of the literature, along with relevant demographic information. One training course run online from the Institute of Statistical Education covered the basics of RMT whilst another in the Netherlands, organised by the experts in the field working under the COSMIN banner, taught me about the relevant measurement properties that a measurement instrument must be shown to have. I also attended the NIHR Academy Members’ Conference in Leeds, which provided training on broader skills like writing and bettering Patient and Public Involvement in research.
As March approached, I was given the green light to begin the data collection but, as we are all aware, this was the beginning of Coronavirus taking a grip across the globe and the necessary lockdowns that came with it. The data collection was put on hold as Covid-19 research became the overriding impetus for all involved with research from both clinical and academic sides. So having been hoping to start in April, and with much rethinking and changes of plan and requests for extensions, there is now the opportunity to start sending out the SOCRATES study packs this month! Within this delay, I have taken the opportunity to engage in even more training courses on RMT and the evaluation of data from PROMs: a further course from the Institute of Statistical Education and another from the University of Western Australia, which were both due to run online. I also joined my supervisor Karl Bang Christensen’s courses from the University of Copenhagen on zoom, but would (of course) much rather have attended them in person as planned.
Dissertation
I also used the time to get a start on writing sections of my dissertation in line with monitoring requirements of my PhD progression. I was able to draft an introduction and start putting flesh to the bones of my systematic review. This systematic review, for which the PROMs discovered from the search are in the data collection, requires the review of articles describing the measurement properties of PROMs measuring RA symptom severity (or disease activity as it is generally described in the literature) in line with the well-defined and recently updated COSMIN guidelines.
I was lucky to be able to present this systematic review to the Outcomes Working Group of the MRC-NIHR Trial Methodology Research Partnership and then gained 4 independent reviewers from across the UK to provide the necessary input to ensure unbiased reviewing of these measurement properties. Many of these independent reviewers have experience of reviewing using the COSMIN guidelines so have added to the methodological aspects of the review, which has led to contact with the COSMIN group to check over various points on how best to review these articles.
Future Plans
As I head into September 2020, I am looking forward to beginning to see data collection forms returning from the persons with RA registered in Cardiff and Vale University Health Board and getting these entered into the database. I am also putting together presentations and posters for the ISOQOL and Health and Care Research Wales Conferences, which are both running virtually and will be a very new experience, along with attending the NIHR Academy Members’ Conference again. I will be attending a new virtual course running from Harvard Chan School of Public Health on ‘Measurement, Design, and Analysis Methods for Health Outcomes Research’ and completing the course from the University of Western Australia. I am pushing forward with completing the article reviews for the systematic review alongside the independent reviewers.
What Links These Studies?
Professor Ernest Choy has provided enormous support throughout my CREATE work, as well as bringing about the SOCRATES application and acting as my primary supervisor. He has been, and currently is, involved in various RA research projects with the Centre for Trials Research. His role as Professor of Rheumatology in the Division of Infection and Immunity within the School of Medicine, and as a Consultant Rheumatologist for Cardiff and Vale University Health Board, puts him in the perfect position to lead and support RA research projects. His experience across clinical and academic fields is vast and he has national and international collaborators to link with as the projects require.
Professor Choy and staff from PRIME and CTR, including Bethan Pell, were involved in the STAR project, which sought to understand the experiences of women with autoimmune rheumatic diseases who were thinking about starting a family, were pregnant or had young children. Details can be found at these links:
http://starfamilystudy.yolasite.com/about-the-study.php
http://www.primecentre.wales/star-family-study.php
Following a successful application to the EU Horizon 2020 funding scheme, Professor Choy and CTR staff David Gillespie, Philip Pallmann and Ian Thomas are collaborating on the DocTIS project, which will see a whole work package running in Cardiff. This project involves collaborators from the UK, Spain, Sweden, Italy, Germany and the USA and aims to identify new combinations of existing drugs that potentiate the effect of each other. This combinatorial effect will have many beneficial aspects for patients: it will lower the disease activity down to remission (i.e. free of symptoms), it will allow to reduce drug dosage (and, therefore, reduce the risk of toxicity) and, finally, it will reduce the probability of becoming resistant to the therapy. More details can be found here http://doctis.eu/
Professor Choy had already formed links with the National Rheumatoid Arthritis Society (NRAS) and this organisation was crucial in my fellowship application: now CEO Clare Jacklin kindly organised a focus group of patients for me to discuss my application with, and I was able to gain their views on the issues that face they face and concerns they had.
It was wonderful to involve Clare on behalf of NRAS in a recent grant application to the NIHR HTA, which involved CTR colleagues and academics from Bangor and Swansea Universities, rheumatologists from across the UK and PPI stakeholders. These PPI stakeholders were the group that I sought out for my fellowship application and formed a vital role in ensuring it had relevance to those that will inevitably be the end users of what this research will create.
Within the Centre for Trials Research, Janine Bates and I have become the co-leads of the Immune Modulated Inflammatory Conditions theme that functions as part of the Infections, Immunity and Inflammation (I3) Group. The CTR I3 Group is overseen by an external panel, which is chaired by Professor Choy. We are excited that September’s CTR I3 Scientific Meeting will focus on Immune Modulated Inflammatory Conditions and we have presentations from both Professor Choy and Bethan Pell to look forward to.
Published Articles (STAR Family Study)
Phillips, R., Pell, B., Grant, A. et al. Identifying the unmet information and support needs of women with autoimmune rheumatic diseases during pregnancy planning, pregnancy and early parenting: mixed-methods study. BMC Rheumatol 2, 21 (2018). https://doi.org/10.1186/s41927-018-0029-4
Phillips R, Williams D, Bowen D, et al. Reaching a consensus on research priorities for supporting women with autoimmune rheumatic diseases during pre-conception, pregnancy and early parenting: A Nominal Group Technique exercise with lay and professional stakeholders. Wellcome Open Research. 2018 ;3:75. https://doi.org/10.12688/wellcomeopenres.14658.1
Williams, D; Webber, J; Pell, B; Grant, A; Saunders, J; Choy, E; Edwards, A; Taylor, A; Wu, M; Phillips, R (2019): “Nobody knows, or seems to know how rheumatology and breastfeeding works”: Women’s experiences of breastfeeding whilst managing a long-term limiting condition- a qualitative visual methods study. Midwifery 78, 91-96. https://doi.org/10.1016/j.midw.2019.08.002
Pell, B; Phillips, R; Williams, D; Sanders, J; Taylor, A; Choy, E; Edwards, A; Grant, A (2020): Using Visual Timelines in Telephone Interviews: Reflections and Lessons Learned from the Star Family Study. International Journal of Qualitative Methods. https://doi.org/10.1177/1609406920913675
- June 2024
- May 2024
- April 2024
- March 2024
- December 2023
- November 2023
- September 2023
- July 2023
- June 2023
- April 2023
- March 2023
- February 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- March 2022
- February 2022
- January 2022
- November 2021
- September 2021
- July 2021
- June 2021
- May 2021
- March 2021
- February 2021
- December 2020
- November 2020
- September 2020
- August 2020
- July 2020
- January 2020
- December 2019
- October 2019
- September 2019
- July 2019
- June 2019
- May 2019
- April 2019
- February 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- December 2017
- October 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- October 2016
- August 2016
- June 2016
- April 2016
- March 2016
- February 2016