Purple Day 2021: Why Epilepsy Research Is So Vital
26 March 2021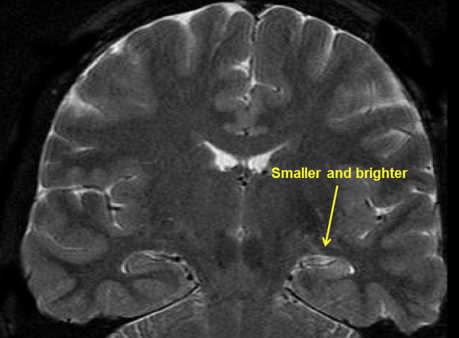
The 26th March is otherwise known as ‘Purple Day’- a day where people are encouraged to where purple in recognition and support of people with epilepsy (PwE).
Epilepsy is the most common chronic neurological condition, affecting between 1-3% of the population. It can affect anyone and although specific epilepsy syndromes ae associated with particular age groups, it can be diagnosed at any time of life. There are over 40 different epilepsy syndromes and seizure types with causative factors including trauma (head injury), brain damage ( from injury or post stroke), development malformations and genetics, plus for a large number of people the underlying cause of their epilepsy may not be known. Therefore, epilepsy is not a heterogeneous condition, leading to challenges in both the understanding of biological mechanisms underpinning seizure activity and in designing research to address potential therapeutic interventions.
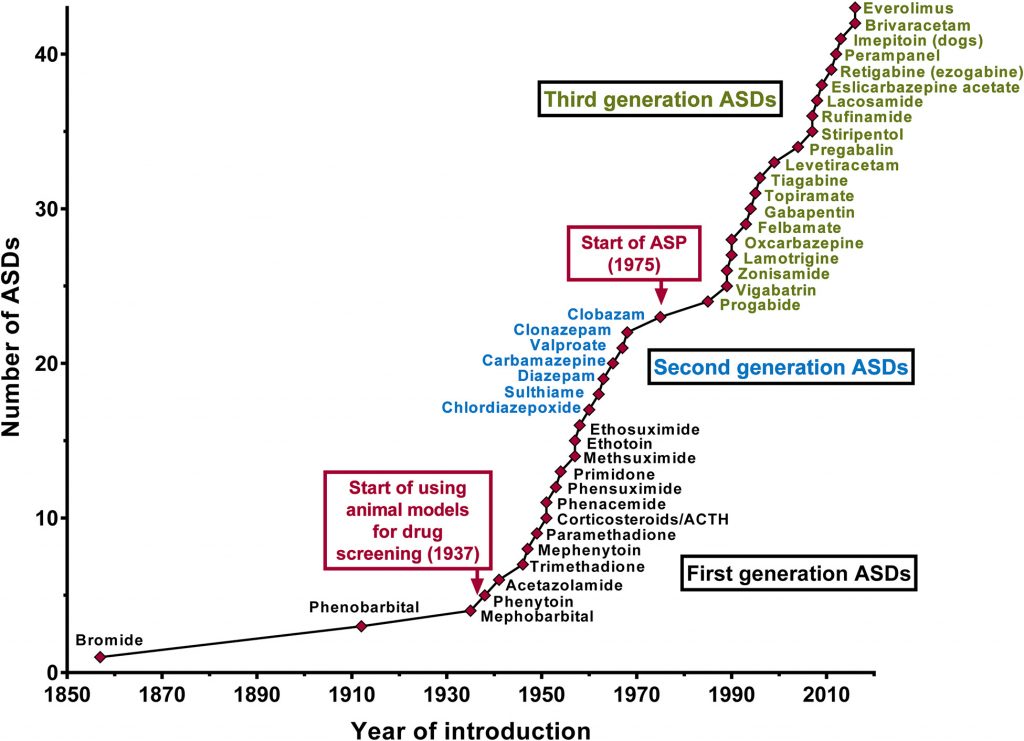
There are a number of medications available to PwE that help prevent their seizures from occurring. However, PwE often have to take more than one medication at different times of day and for up to 30% of PwE, current medications do not work to control their seizures (known as intractable or drug resistant epilepsy). There have been real advances over the last 30 years or so with the introduction of novel anti-convulsant medications identified through the Anti-convulsant screening program (ASP) and improvements in surgical techniques afford those with a clear epileptic focus in a ‘non-essential’ brain region the opportunity for potential seizure freedom through removal of the epileptiform brain tissue. However – for those where drugs don’t work and surgery is not an option, there is still a real need for novel treatments for improving seizure control.
In 2009, the Wales Epilepsy Research Network partnered with the James Lind Alliance to identify the research questions most important to PwE, epilepsy clinicians and epilepsy researchers. Interestingly, the results were largely skewed towards the understanding of living with epilepsy and how treatment impacts daily living. This serves to highlight that even though outwardly, seizure control is paramount, the complexities of living with epilepsy and taking daily medication have long-term and significant impacts on PwE, which they themselves want to understand better and to be addressed by high quality research. It is clear that social stigma is still prevalent among PwE
Some of the work we have done here in Cardiff around South Wales, has been aimed at investigating these about aspects of quality of life and living with epilepsy with a view to improving treatment, clinical care and social support for PwE. Prof Khalid Hamandi currently holds an Epilepsy Research UK project grant to use advanced imaging methods (fMRI and Magnetoencephalography [MEG]) to generate non-invasive brain mapping of epileptic activity to improve predicted outcomes in epilepsy surgery and to be able to offer surgery as a therapeutic option to a wider range of PwE.
Another study run here in Cardiff is FLAME. This was a Phase II, double blind randomised controlled trial looking at the feasibility of using fluoxetine (more commonly known as Prozac) for treating learning and memory dysfunction in people with temporal lobe epilepsy (TLE), where problems with learning and memory are reported by people with TLE as having a major impact on their quality of life. TLE describes epilepsy where the seizure focus is located in the temporal lobe and is often associated with hippocampal sclerosis (which describes a loss of cells in the hippocampus, the brain region known to be important for spatial learning and memory and can be seen in the MRI image below).

We know that the hippocampus is a brain region where new brain cells are generated throughout life (a process known as neurogenesis) and that this process can be manipulated by pharmacotherapy. Pre-clinical research using an animal model of TLE demonstrated a lack of neurogenesis, but could be rescued by fluoxetine, which is known to stimulate neurogenesis. The next step was to then test this in a clinical setting.
Unfortunately, the trial experienced significant issues with recruitment and was found to be unfeasible in the existing format. Given the significant issues experienced with recruitment, we undertook a post-hoc process evaluation to better understand the barriers and reasons for non-participation. The main reasons that emerged were stigma associated with taking fluoxetine, the need to take time off work and principally that people with good seizure control did not want to risk the potential of disturbing that by taking a new medication; this centres around the potential loss of driving ability and hence personal freedoms, and ability to work.
Those working in clinical research know that recruitment is always hard, but for these and other reasons, recruitment into epilepsy research appears to be harder. Work from Swansea University and using the SAIL databank showed that the incidence of epilepsy in Wales is associated with socio-economic deprivation and one might expect that this would bear relation to the opportunities and accessibility of PwE in these geographical areas for taking part in research. However, there are still many aspects about the barriers to participation that we don’t fully understand. In attempt to combat this, we are in the process of delivering a Public Engagement (EMPOWER) initiative in these areas in an effort the to improve the understanding and accessibility of clinical research to PwE in Wales.
Whilst there is ongoing research to; a) better understand how the brain generates epileptiform activity (see work by Vincenzo Crunelli in School of Biosciences) to improve our understanding of how we might combat the origins of seizure activity and b) generate novel ways of interrupting seizure networks through gene therapy approaches and optogenetics (using light to control neurons that have undergone genetic manipulation) there is still a pressing need to conduct research into how we can improve living with epilepsy for PwE and better design clinical studies that are inclusive of those PwE most in need.
- June 2024
- May 2024
- April 2024
- March 2024
- December 2023
- November 2023
- September 2023
- July 2023
- June 2023
- April 2023
- March 2023
- February 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- March 2022
- February 2022
- January 2022
- November 2021
- September 2021
- July 2021
- June 2021
- May 2021
- March 2021
- February 2021
- December 2020
- November 2020
- September 2020
- August 2020
- July 2020
- January 2020
- December 2019
- October 2019
- September 2019
- July 2019
- June 2019
- May 2019
- April 2019
- February 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- December 2017
- October 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- October 2016
- August 2016
- June 2016
- April 2016
- March 2016
- February 2016