Better Healthcare Through More Inclusive Research – A Dissemination Event On Innovations In Clinical Trials Design And Delivery For Under-Represented Groups
13 December 2018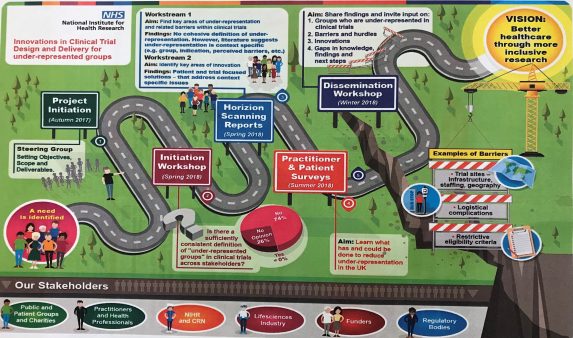
A vision for ‘better healthcare through more inclusive research’
During the past year, a project looking at innovations in clinical trials design and delivery for under-represented groups has been carried out by a group of researchers at Newcastle University as part of a NIHR CRN speciality cluster, and supported by the NIHR Innovation Observatory. The project consisted of two horizon scanning reviews, a practitioner and patient survey, and engagement with a range of stakeholder groups. A Steering Group was involved in setting the objectives, scope and deliverables.
A dissemination workshop was held in London (December 2018) to share the findings, and invite input from the wider researcher and practitioner community, which I was fortunate to be able to attend. After an introduction from the project lead Professor Lynn Rochester, a number of areas were explored using the findings from the reviews and survey, and experiences from different perspectives.
Why is inclusive research important?
Ensuring research is inclusive means that all members of the population have an evidence base for the treatment and care they receive. For example, a treatment or intervention that is effective in a clinical trial of young and healthy individuals may not be effective in older people with multi-morbidity, or worse it may actually be harmful. It is also important that we are not excluding people from research, in the same way we shouldn’t be excluding people from accessing any services or treatment by virtue of them being a member of a minority group or living with a disability.

What do we understand by ‘under-represented’ groups?
The first question of the day was to define and understand the meaning of under-represented groups in clinical trials. The first review had identified a number of groups from the literature such as minority groups, older people (particularly those living with frailty and multi-morbidity), and those from more socio-economically deprived populations. Although it was stressed that it is a term that lacks definition, and is very much related to the context of the clinical trial and populations involved.

The survey findings suggested that there are other groups that may also be considered to be currently under-represented in clinical trials.

A later part of the event looked at what terms could be used as an alternative to ‘under-researched’ that are more inclusive, using small-group discussions and interactive methods to explore delegates’ views and suggest alternatives.
Key areas of innovation to address under-representation
The second review presented aimed to identify key areas of innovation reported in the literature. These were both patient-focused and trial-focused solutions to inclusion, such as inclusive trial design and modernising inclusion eligibility.

Successful innovations identified from the survey were those such as more inclusive recruitment and consent procedures, and ways of working that value people from minority communities.
Learning from experience
Three case studies presented valuable experiences from different perspectives. An insight into the participant perspective was provided by Francine and Cath who spoke about their experiences of engaging with (and being part of) a marginalised population, together with Professor Fiona Nolan. Wajid Kahn talked about the experience of engaging with under-represented populations as part of research delivery. Professor Miles Witham spoke about the importance of inclusion and the impact on the available evidence base, both a researcher and a clinician.

The main messages were that communication and building relationships are key, trials need to be thoughtfully designed to take account of under-representation and address any barriers, greater support from funders is crucial, and that more research is needed on how we can make research more inclusive.
Next steps and a prioritised call to action
The day ended with a series of interactive questions posed to delegates, including identifying what the priorities are to achieve the aim of better healthcare through more inclusive research. There was a wide variety of views from across the groups, including that better engagement outside of healthcare services (including with social care) was needed. I look forward to hearing more about the findings and how we, as researchers, can better design inclusivity into clinical trials and other research studies.
Event Hashtag: #RepresentationInResearch
- June 2024
- May 2024
- April 2024
- March 2024
- December 2023
- November 2023
- September 2023
- July 2023
- June 2023
- April 2023
- March 2023
- February 2023
- December 2022
- November 2022
- October 2022
- September 2022
- August 2022
- July 2022
- June 2022
- May 2022
- April 2022
- March 2022
- February 2022
- January 2022
- November 2021
- September 2021
- July 2021
- June 2021
- May 2021
- March 2021
- February 2021
- December 2020
- November 2020
- September 2020
- August 2020
- July 2020
- January 2020
- December 2019
- October 2019
- September 2019
- July 2019
- June 2019
- May 2019
- April 2019
- February 2019
- December 2018
- November 2018
- October 2018
- September 2018
- August 2018
- July 2018
- June 2018
- May 2018
- April 2018
- March 2018
- December 2017
- October 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- October 2016
- August 2016
- June 2016
- April 2016
- March 2016
- February 2016